The future of pharmaceutical documentation is here – faster, smarter and compliant. That’s what World Pharma Today learnt from its recent interview with Docuvera. Striking the right chord between regulatory compliance and automation, Docuvera has indeed changed the way pharmaceutical documentation is being analyzed and executed.
1. How does Docuvera’s AI-driven approach transform the erstwhile traditional pharmaceutical documentation process into a more error-free and efficient workflow?
Docuvera’s AI-driven approach transforms traditional pharmaceutical documentation by uniquely combining AI with structured content methodologies. Unlike typical Generative AI solutions, which primarily generate new content and necessitate extensive review, Docuvera prioritizes content reuse strategies over traditional GenAI.
- RARe (Retrieval-Augmented Reuse): This method involves directly reusing validated, human-reviewed content components. Since these components have already undergone thorough human review, it significantly reduces or eliminates the need for subsequent review cycles, thereby enhancing efficiency and reducing the risk of errors.
- RAT (Retrieval-Augmented Transformation): RAT involves slight modifications or transformations of existing human-reviewed content. The minor nature of these changes means that review cycles are minimal, as reviewers can quickly assess and verify these incremental adjustments, thus maintaining accuracy with improved efficiency.
- RAG (Retrieval-Augmented Generation): When existing content is unavailable or insufficient, RAG is employed to generate new content using validated examples of human-reviewed components. Although this approach creates fit-for-purpose content, it inherently requires additional review to confirm accuracy and relevance. Thus, RAG, while effective, is positioned last due to its comparatively higher review and validation requirements.
By prioritizing these methods, Docuvera ensures that pharmaceutical documentation processes become significantly more efficient, reducing manual review burdens and minimizing potential errors.
2. Can you share some insights on how AI has practically helped reduce manual effort and simultaneously be very exact in pharmaceutical documentation?
Regulatory labeling is one example where AI has significantly streamlined pharmaceutical documentation. For example, a user can efficiently generate a new Core Data Sheet (CDS) and regional labels by employing Docuvera’s prioritized methodologies:
- RARe: Users can directly reuse content from clinical documents and previously approved CDSs to swiftly build a robust first draft of a new CDS, eliminating the time-intensive task of manual content creation and initial validation.
- RAT: Minor content transformations—such as updating product names—can be performed quickly while preserving most of the already validated content. This minimal alteration drastically shortens review cycles, ensuring accuracy is maintained without extensive re-validation.
- RAG: When content needs exceed available validated materials, new content can be efficiently generated by leveraging relevant examples from clinical and regulatory documents, as well as publicly available sources like DailyMed. Although RAG-generated content requires additional review, the AI ensures the generated material aligns closely with established content standards, simplifying the validation process.
Once the CDS is complete, users can further leverage RARe, RAT, and RAG methods to efficiently create regional and local labels, such as US Prescribing Information (USPI) and the Summary of Product Characteristics (SmPC). This involves reusing validated content directly from the CDS (RARe), making necessary minor transformations to align with regional regulatory requirements (RAT), and generating new, region-specific content when necessary, using the CDS content as a reference model (RAG). This approach ensures consistency, accuracy, and compliance while substantially reducing manual effort and review cycles across diverse regional documentation.
3. How does Docuvera’s approach strike the right balance between automation and governance to ensure regulatory compliance in the highly complex pharmaceutical landscape?
Docuvera’s approach adeptly balances automation and governance, essential for regulatory compliance in the highly complex pharmaceutical industry. By integrating AI-driven content management with structured governance frameworks, Docuvera provides essential safeguards such as:
- Robust audit trails: Comprehensive records of all changes made, capturing who made them, when, and why, ensuring transparency and traceability.
- Detailed version control: Precise management of document and component revisions, maintaining clarity about document history and allowing easy retrieval of specific versions.
- Clear distinctions between AI-generated and human-reviewed content and identification of content sources: Transparent identification of the state of content (has the content been human reviewed?) and the content sources from which the content was derived. This information allows reviewers and approvers to focus their oversight efforts in the most appropriate areas.
- Review and approval workflows: Integrated workflows involving multiple review stages by subject matter experts and compliance officers, guaranteeing thorough vetting of documentation.
- Clear permissions model: Defined access controls based on roles, ensuring only authorized individuals can generate, reuse, access and modify content, safeguarding against unauthorized changes.
- Software validation: Rigorous testing and validation of AI tools and software to confirm accuracy, reliability, and compliance with regulatory standards, essential for trustworthy and compliant documentation outputs.
These elements collectively ensure transparency, accountability, and traceability, which are critical for maintaining regulatory compliance.
4. Following regulations holds the key to success for any pharmaceutical setup. How does the use of artificial intelligence ensure proper document-updation so as to meet the evolving regulatory needs of the sector?
Docuvera leverages artificial intelligence and structured content methodologies to ensure documents continuously meet evolving regulatory needs:
- Within structured content, templates can be modified (structure, formatting, metadata) to ensure that new documents always align with the latest regulatory guidelines.
- Changes made to existing documents can efficiently propagate using structured content’s change propagation feature, where updates in reference documents cascade to all documents reusing that content, ensuring uniform compliance.
- AI tools can be infused with the latest regulatory guidance, automatically checking content to verify compliance with current standards, thereby significantly reducing compliance risks and enhancing the speed and accuracy of document updates.
This approach ensures that regulatory intelligence is consistently injected into content creation, updates, and review cycles.
5. How can the AI-led tools help in pharmaceutical companies accelerate drug development timelines without compromising on the quality of the document?
The combination of AI and Structured Content within Docuvera accelerate pharmaceutical documentation timelines significantly, without compromising document quality, through several focused efficiencies:
- Faster creation of accurate draft content through prioritized use of RARe, RAT, and RAG methodologies, substantially reducing initial drafting time.
- Dramatic reduction in review cycles by reusing previously validated human-approved content, minimizing the need for extensive re-validation.
- Efficient reuse of content from reference documents enables the rapid creation of new derivative documents, maintaining consistency and compliance.
- Rapid propagation of changes from source content to all reused instances ensures timely and consistent updates while reducing errors and omissions.
- Reduction of errors and omissions by reusing pre-approved, locked content components, rather than generating entirely new content, thereby ensuring greater accuracy and compliance.
6. How can structured content management make sure of consistency of content across numerous types of pharmaceutical documents such as a drug evaluation report or a clinical trial submission data?
Structured content management ensures consistency across numerous pharmaceutical document types, such as drug evaluation reports and clinical trial submissions, by:
- Enabling direct reuse of validated, human-reviewed content components, ensuring uniformity and consistency.
- Utilizing minor content transformations (RAT) to align reused content precisely to specific requirements of new documents.
- Facilitating efficient propagation of updates from source content to derivative documents, maintaining accuracy and compliance throughout document lifecycles.
- Reducing the potential for errors by employing previously validated and locked content components, which minimizes discrepancies and ensures high-quality, consistent documentation.
- Content validation tools can then check content to ensure it is fit-for-purpose while identifying content non-conformities that need to be addressed
7. Are the AI tools that Docuvera implements in report generation capable enough to gauge a potential compliance risk before the document gets submitted?
Docuvera is implementing advanced content integrity tools infused with the latest regulatory intelligence, capable of proactively checking pharmaceutical documents to ensure completeness and accuracy. These tools automatically detect potential compliance risks, such as missing or inconsistent information, before document submission. By continuously updating with current regulatory standards, the AI tools help pharmaceutical companies identify and address potential compliance issues early, significantly reducing the risk of regulatory non-compliance and enhancing the reliability and quality of the submitted documentation.
8. When we talk of large-scale global pharma giants, collaboration between cross- functional teams is pivotal. How does Docuvera’s platform help in ensuring that seamlessness.
Docuvera supports seamless collaboration across cross-functional teams within large-scale global pharma organizations by providing an enterprise-wide solution rather than a limited point solution. It is configurable to effectively address multiple pharmaceutical domains including Clinical, Regulatory Labeling, CMC, Medical Affairs, and Safety use cases.
Content developed within these domains can be efficiently reused across teams by employing RARe, RAT, and RAG methodologies, enabling rapid generation of high-quality, consistent new documents. Additionally, Docuvera’s workflows facilitate simultaneous collaboration, allowing multiple team members across different departments to concurrently review, update, and approve documents.
A robust Role-Based Access Control (RBAC) model ensures security and compliance, precisely managing permissions for document access, creation, and modification, ensuring only authorized personnel participate in sensitive processes. Comprehensive audit trails and detailed versioning further enhance transparency and accountability, fostering effective collaboration and alignment among cross-functional teams, thus optimizing productivity and regulatory compliance.
9. What are further steps that are being taken by Docuvera to integrate emerging AI tech so as to further enhance documentation?
Docuvera is actively expanding its AI integration strategy with several key initiatives planned for implementation, e.g. layering advanced technologies on top of the structured content architecture to enhance documentation processes across the pharmaceutical lifecycle:
- Enhancing our GenAI capabilities beyond the current implementations for domain-specific content generation and auto-propagation
- Expanding Natural Language Processing applications beyond the existing topic classification for IDMP submissions
- Advanced Machine Translation (MT) capabilities for primary and back translations of medical content
- Enhanced Machine Learning (ML) algorithms to analyze content architecture and improve search relevance fundamental to RARe
- Content integrity verification based on Regulatory Intelligence leveraging NLP and ML to ensure document completeness and accuracy
- Workflow optimization using ML to suggest appropriate reviewers and approvers based on domain expertise and workload
10. How does Docuvera ensure that the structured content management is user-friendly and can be embraced by even the non-tech personnel?
Docuvera’s structured content management system is designed with user-friendliness as its primary guiding principle. The platform combines a familiar, modern interface with powerful structured authoring capabilities that don’t require technical expertise to use effectively.
The user interface operates similarly to Microsoft Word, allowing non-technical users to create and edit documents in a single view without needing to understand complex XML tagging or technical structures. Users can navigate to sections and add paragraphs, tables, figures, or equations just as they would in familiar word processing applications. This Word-like experience has driven strong adoption rates across global implementations.
The system handles the technical aspects of structured content management behind the scenes, allowing authors to focus on content rather than formatting or technical requirements. And, with AI layered on top of our intuitive interface, many of the manual processes required in Structured Content are replaced by automated activities (e.g., content generation, reuse, reviews, etc.).
11. AI-powered structured content management from Docuvera delivers documented110% productivity gains and 145% ROI when it comes to pharmaceutical documentation. Can you elaborate on these factual representations?
As content is effectively reused:
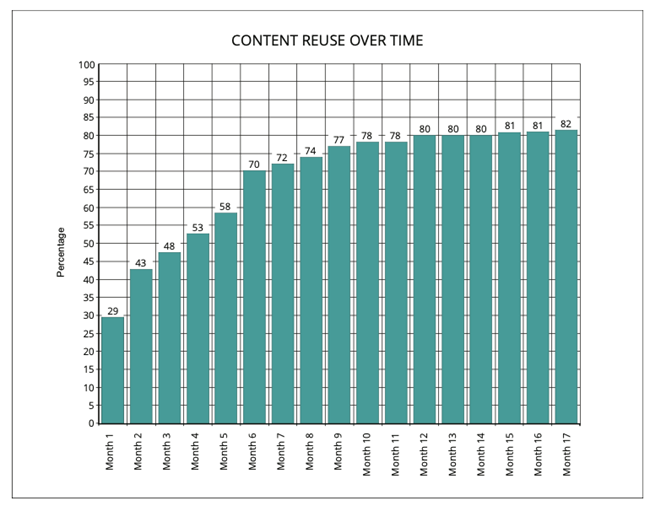
The number of documents in which a user can be involved increases:
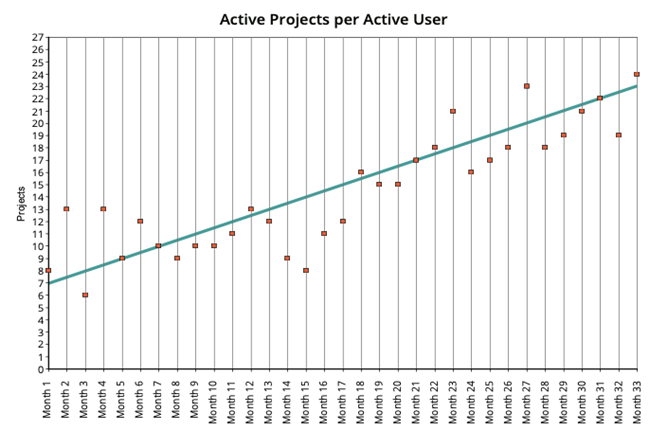
Let us know if you need any more details on this.






















