TraceLink’s Global Drug Supply, Safety and Traceability Report
survey highlights a lack of industry readiness for U.S. DSCSA, and
concerns in European nations on readiness for the February 2019
EU FMD compliance deadline
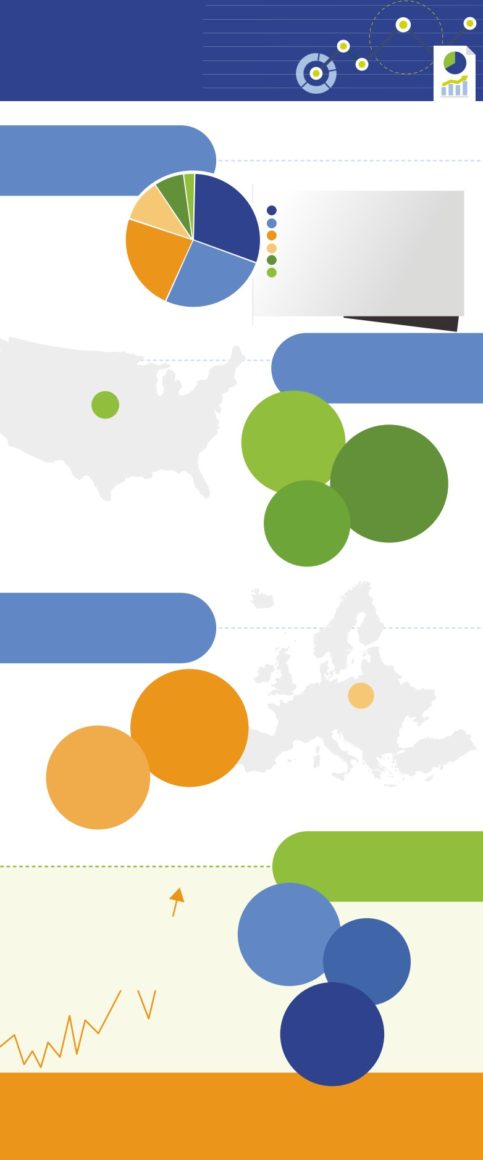
Organization Breakdown*
2%
7%
12%
30%
Hospitals n=195
Pharmaceutical companies n=174
Contract Manufacturing Organizations (CMOs) n=155
Respondents
Pharmacies n=81
Wholesale Distributors n=50
surveyed
23%
Third Party Logistic Companies n=11
660
26%
*all with compliance regulations for either the Food and Drug
Administration’s (FDA) Drug Supply Chain Security Act (DSCSA) and/or the
European Union’s Falsified Medicines Directive (FMD)
U.S. Serialization Readiness:
33%
Industry Belief vs. Reality
of pharma
companies
believe
VS.
they are
25%
BUT
“very ready”
“actually ready”
not one
for serialization
based on
pharma company is
necessary steps
100%
taken
ONLY 11%
ready
of pharma companies
feel that their CMOs
were ready to ship
50%
serialized products
CMOs that feel
Utmost Concern:
“very ready”
for serialization
98% of CMOs had concerns that equipment
shortages will affect customer compliance
EU Serialization Readiness:
Industry Belief vs. Reality
33%
of pharma
companies feel
“very ready”
4%
for serialization
VS.
ONLY 15%
EU pharma companies
of EU pharma
believe they will be
companies and
sending serial numbers
CMOs are truly
to the EU Hub in time for
on target for
the February 2019
serialization
NOT ONE
deadline
readiness
EU pharma company is
integrated with the
majority of their CMOs
Hidden Values Behind
Serialized Data
Product
authentication
Majority of respondents
and secure
Inventory
see added value in
distribution
and recall
serialized data
management
Better business
relationships up and
down the drug
supply chain
Networked ecosystems of trading partners
stand to achieve the most significant ROI
from serialization data
To view the entire report visit: https://www.tracelink.com/global-drug-supply-report/2017-report

















![Sirio Launches Global Research Institute for Longevity Studies [SIA]](https://www.worldpharmatoday.com/wp-content/uploads/2019/09/Sirio-218x150.jpg)

